

Next: Solution Free Energies that
Up: Lecture_31_web
Previous: Lecture_31_web
Phase diagrams have been constructed for the case
of one component (
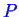 -
-
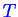 diagrams for a pure material),
and for two component systems (
diagrams for a pure material),
and for two component systems (
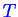 -
-
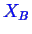 diagrams drawn
at constant pressure).
Each time a new component is added, another intensive variable
must be held constant if the phase diagram is to be drawn in
two-dimensions.
diagrams drawn
at constant pressure).
Each time a new component is added, another intensive variable
must be held constant if the phase diagram is to be drawn in
two-dimensions.
For ternary systems, there are three components. Let
the three components be denoted by
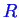 ,
,
 , and
, and
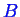 .
Because,
.
Because,
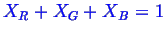 , the system can be
represented by two components, say
, the system can be
represented by two components, say
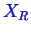 , and
, and
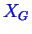 , and
the phase diagram could be represented in the following
coordinate system:
, and
the phase diagram could be represented in the following
coordinate system:
Figure 31-1:
Possible way to draw a ternary phase diagram at constant pressure.
It would be difficult to interpret such diagrams.
 |
Question: what is the maximum number of
phases that can be in equilibrium at one
point in Figure 31-1?
It may be possible to represent such a diagram in two dimensions
by taking slices at constant composition, for instance:
Figure 31-2:
Pseudo-binary slices of a ternary phase diagram at constant pressure.
The figure on the left is a true binary phase diagram and has the same corresponding rules
for the degrees of freedom.
 |
Ternary phase diagrams are traditionally drawn at constant pressure
and temperature--and the following scheme is used to represent all
three components:
Figure 31-3:
Representation of three components at constant
pressure and temperature. Each triangle vertex corresponds to a pure component.
Each triangle side corresponds to: 1) a system with none of the component
from the opposite vertex; 2) a binary alloy with none of the third component
represented by the opposite vertex.
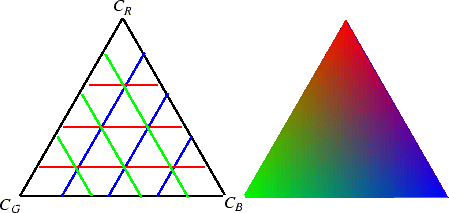 |
For example, a ternary phase diagram may look something like this:
Figure 31-4:
An example of a ternary phase diagram.
Three phase regions become triangles where the limiting
composition of each co-existing phase is given by the
vertices of the triangle.
The sides of the triangle are the limits of the tie-lines from
an abutting two phase region.
The lever rule in three phase region is graphically illustrated by
the weighted phase fractions distributed about the average composition.
 |
Figure 31-5:
Example of a simple ternary phase diagram
at constant  and
and 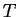 .
.
 |


Next: Solution Free Energies that
Up: Lecture_31_web
Previous: Lecture_31_web
W. Craig Carter
2002-12-03
![]() -
-
![]() diagrams for a pure material),
and for two component systems (
diagrams for a pure material),
and for two component systems (
![]() -
-
![]() diagrams drawn
at constant pressure).
Each time a new component is added, another intensive variable
must be held constant if the phase diagram is to be drawn in
two-dimensions.
diagrams drawn
at constant pressure).
Each time a new component is added, another intensive variable
must be held constant if the phase diagram is to be drawn in
two-dimensions.
![]() ,
,
![]() , and
, and
![]() .
Because,
.
Because,
![]() , the system can be
represented by two components, say
, the system can be
represented by two components, say
![]() , and
, and
![]() , and
the phase diagram could be represented in the following
coordinate system:
, and
the phase diagram could be represented in the following
coordinate system:



