

Next: About this document ...
Up: Lecture_26_web
Previous: Various Confusing Issues on
When there is only one degree of freedom in a single component
phase diagram, it was shown above that there
must be a relation between
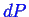 and
and
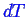 for the
system to remain in two phase equilibrium.
Such a relation can be derived as follows:
for the
system to remain in two phase equilibrium.
Such a relation can be derived as follows:
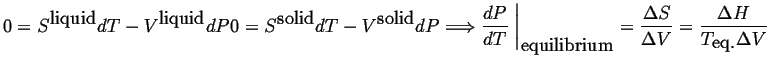 |
(26-2) |
Equation 26-2 is the famous Clausius-Clapeyron
equation.
Consider the behavior of the molar free energy (or
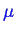 ) on slices of Figure 26-1
at constant
) on slices of Figure 26-1
at constant
 and
and
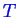 :
:
Figure 26-2:
Considerations of the molar Gibbs free
energy on slices of the single component phase diagram
along lines of constant 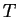 and constant
and constant 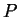 .
.
 |
Figure:
Behavior of
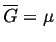 at constant
at constant 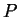 as a function of
as a function of 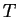 .
Where the curvature of
.
Where the curvature of
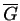 changes sign, the system is unstable.
The liquid and vapor curves must be connected to each other and this
is illustrated with the "spiny-looking" curve with opposite curvature.
The curve for solid is not connected to the others.
changes sign, the system is unstable.
The liquid and vapor curves must be connected to each other and this
is illustrated with the "spiny-looking" curve with opposite curvature.
The curve for solid is not connected to the others.
 |
Figure:
Behavior of
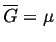 at constant
at constant  as a function of
as a function of  .
.
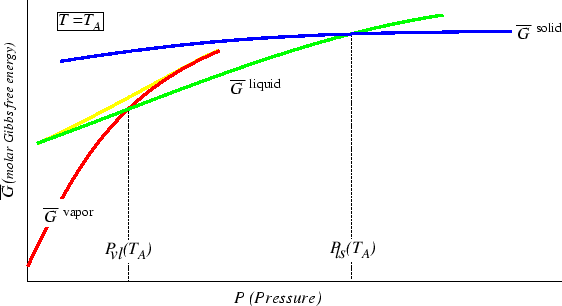 |
Figure 26-5:
Example of single component phase diagram plotted
with
one derived intensive variable.
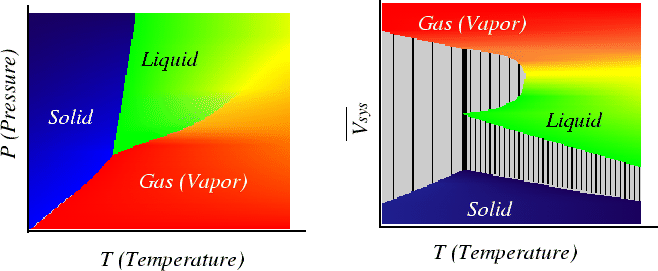 |
What would the plot look like with two extensive variables plotted?


Next: About this document ...
Up: Lecture_26_web
Previous: Various Confusing Issues on
W. Craig Carter
2002-11-21
![]() and
and
![]() for the
system to remain in two phase equilibrium.
Such a relation can be derived as follows:
for the
system to remain in two phase equilibrium.
Such a relation can be derived as follows:
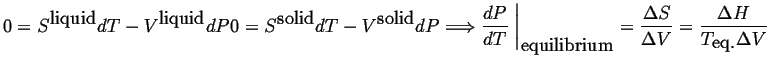
![]() ) on slices of Figure 26-1
at constant
) on slices of Figure 26-1
at constant
![]() and
and
![]() :
:

