| (26-1) |
| (26-1) |
The Gibbs phase rule is a very useful equation because
it put precise limits on the number of phases
![]() that
can be simultaneously in equilibrium for a given number
of components.
that
can be simultaneously in equilibrium for a given number
of components.
What does Equation 26-1 mean? Consider the
following example of a single component (pure) phase
diagram
![]() .
.
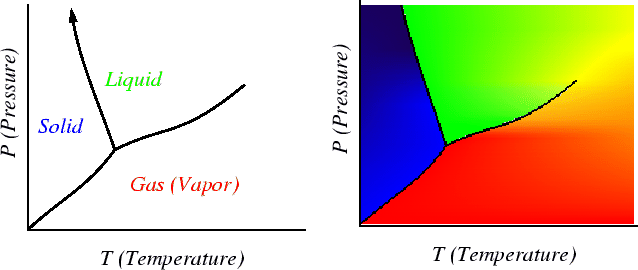 |
Consider a single-phase region:
![]()
This implies that two variables (
![]() and
and
![]() ) can
be changed independently (i.e.,
pick any
) can
be changed independently (i.e.,
pick any
![]() and
and
![]() ) and a single phase remains in
equilibrium.
) and a single phase remains in
equilibrium.
Consider where two phases are in equilibrium:
![]() ,
,
There is only one degree of freedom-for the two
phases to remain in equilibrium, one
variable can be changed freely (for instance,
![]() ) but then
the change in the other variable (i.e.,
) but then
the change in the other variable (i.e.,
![]() ) must depend
on the change of the free variables:
) must depend
on the change of the free variables:
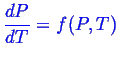
Finally, consider where three phases are in
equilibrium then:
![]() .
.
There can be no change any variable that maintains three phase equilibrium.