It can be shown that the ideal solution represents the limiting behavior of very dilute solutions. The question may be posed: ``In what cases can we expect the activity to depend only on composition?''
Consider a very dilute solution of
![]() in
in
![]() :
:
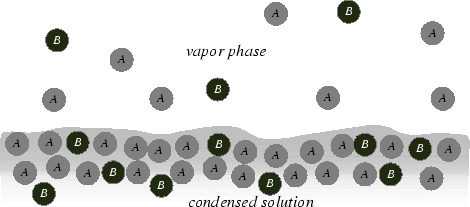 |
Each time an
![]() comes out of solution, it
does so mostly without any influence of
comes out of solution, it
does so mostly without any influence of
![]() .
It is as if it does so as in a pure solution.
.
It is as if it does so as in a pure solution.
Each time a
![]() comes out of solution, it
does so entirely under the influence of
the surrounding
comes out of solution, it
does so entirely under the influence of
the surrounding
![]() atoms; it is as if it does so from pure
atoms; it is as if it does so from pure
![]() .
.
So one can expect very concentrated, or very dilute solutions to behave ideally.
Typically, the data look like the following:
One gets as limiting behavior:
Raoult's Law:1
| (31-5) |
and Henry's law:
| (31-6) |