 |
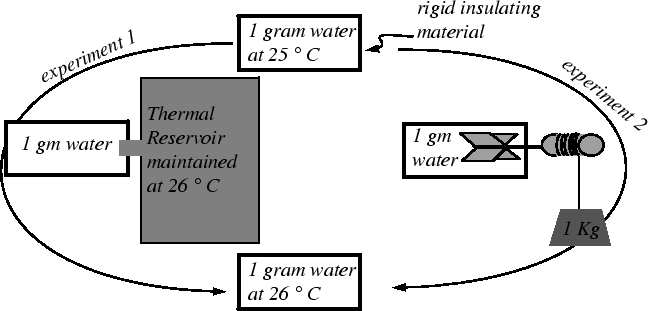
Consider again the classical experiment that illustrates the equivalent effects of adding heat to a system and doing an equivalent amount of work on a system.
 |
The final conclusions of Joule's paper were:
That the quantity of heat produced by the friction of bodies, whether solid or liquid, is always proportional to the quantity of force extended.
That the quantity of heat capable of increasing the temperature of a pound of water (weighed in vacuo, and taken between 55and 60
F) by 1
F requires for its evolution the expenditure of a mechanical force represented by the fall of 772 lb. through a space of one foot.
This states that work, defined here as a mechanical force exerted over a displacement, can be converted into an equivalent amount of heat.
Question: How long would it take to heat water about 5 degrees F?
We can state the first law as follows:
| First Law of Thermodynamics: Version 1 |
| A state function, called the internal energy, exists for any physical system--and the change in the internal energy during any process is the sum of the work done on the system and the heat transfered to the system. |
There are several interpretations of the first law:
And, now for something completely different:
How could I have have doubted your constancy
your warmth and labors summed, signify
your ever unchanging incessancy.
Though some virtues wane, will others supply
perfect completeness in my universe.
Are we not a pair, born of heat and toil,
controvertable, not better nor worse.
Though shoulders may be cold or blood may boil,
you are balanced and never violated.
Timeless compass of perfect reckoning,
northen star of mislaid paths translated,
you, the first, though a second, beckoning
Nature, forever, through her laws eschew
is
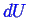
W. Craig Carter, 2002
To define work, we need to consider all of the possible ways
that we can change the extent of a system.
Let
![]() represent the change in some arbitrary extensive quantity (volume,
charge, number of atoms of a certain type, total magnetization, etc)
of a system and suppose that we construct some mechanical device to produce that
change--just like in Joule's experiments.
Let the force be exerted by the device--during the change
represent the change in some arbitrary extensive quantity (volume,
charge, number of atoms of a certain type, total magnetization, etc)
of a system and suppose that we construct some mechanical device to produce that
change--just like in Joule's experiments.
Let the force be exerted by the device--during the change
![]() -be
-be
![]() , then
the rate at which work is done per change
, then
the rate at which work is done per change
![]() is
is
![]() .
If there are
.
If there are
![]() different extensive quantities (
different extensive quantities (
![]() ,
,
![]() , ...,
, ...,
![]() )
then the rate at which work is done is:
)
then the rate at which work is done is:
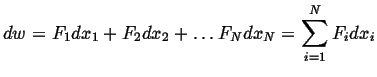 |
(05-1) |
Important: the
![]() refer to the device that is producing the extent
refer to the device that is producing the extent
![]() , while
the
, while
the
![]() refer to the system's extensive variables.
refer to the system's extensive variables.
Question: Suppose the system changes from some initial state
![]() to some final state
to some final state
![]() ,
could we just integrate to find:
,
could we just integrate to find:
 |
(05-2) |
Because the forces refer their values during a change in a system, we can write the first law as a differential expression:
| Differential Form of the First Law of Thermodynamics |
|
|
| Those Pesky Minus Signs | ||
| First Law Form | Work | Heat |
|
|
||
|
|
||
|
|
||
|
|
||
Let's work out a simple example for a capacitor:
Examples of different ways that work can be done will appear in later lectures.
For the definition of heat, let's use the definition of work and the first law and define heat is the work-less transfer of internal energy from one system to another. From our definition of the Zeroth law, it follows that heat flows from higher to lower temperature systems. To repeat, temperature refers a property of a system while heat refers to a process--the process of exchanging that part of internal energy that is not associated with work.